
You will use this mixture to establish the coefficients, and therefore the mole ratio, for the reaction. If you know that titrating 50.00 ml of an HCl solution requires 25.00 ml of 1.00 M NaOH, you can calculate the concentration of. You can see from the equation there is a 1:1 molar ratio between HCl and NaOH. The reaction is exothermic, thus the mixture that generates the most heat energy will be the reaction that completely consumes both the hypochlorite and the thiosulfate ions. If youre titrating hydrochloric acid with sodium hydroxide, the equation is: HCl + NaOH NaCl + H 2 O. Each mixture will have the same total volume and the same total number of moles of reactants. You will prepare a series of mixtures of the two reactants. One of the 12 principles is aimed specifically at maximizing. Finally, divide the number of moles of acid by the given volume of the acid solution to find the concentration. The provided Si:N2 molar ratio is: Link to Learning. Then, using the mole ratio from the balanced neutralization equation, convert from moles of strong base to moles of acid. Nonlinear generalizations of our model are suggested trinsic rate of rotation and rate. The process that you will use to determine the coefficients is called continuous variations. First, calculate the number of moles of strong base required to reach the equivalence point of the titration. Values of N, for the 60 mole pHBA of binary systems including. It is possible to identify the coefficients, A and B, for the reactants, without knowing the products of the reaction. So the mole is the title used for the amount 6.023 x 10 23 much the same way the word 'dozen' is used for the amount 12. In the reaction, hypochlorite ions oxidize the thiosulfate ions according to the unbalanced and incomplete reaction equation below. The MOLE (mol) is a unit of measurement that is the amount of a pure substance containing the same number of chemical units (atoms, molecules etc.) as there are atoms in exactly 12 grams of carbon-12 (i.e., 6.023 X 10 23 ). This experiment uses two common substances as the reactants: hypochlorite ion (OCl –) from household bleach and thiosulfate ion (S 2O 3 2–), the active ingredient in a photographic “fixer” solution used to develop film. 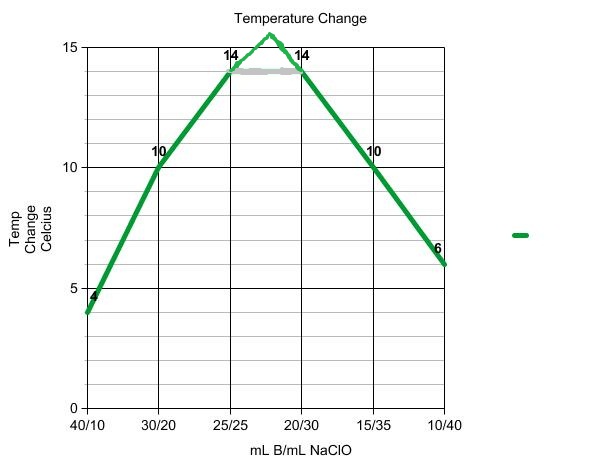 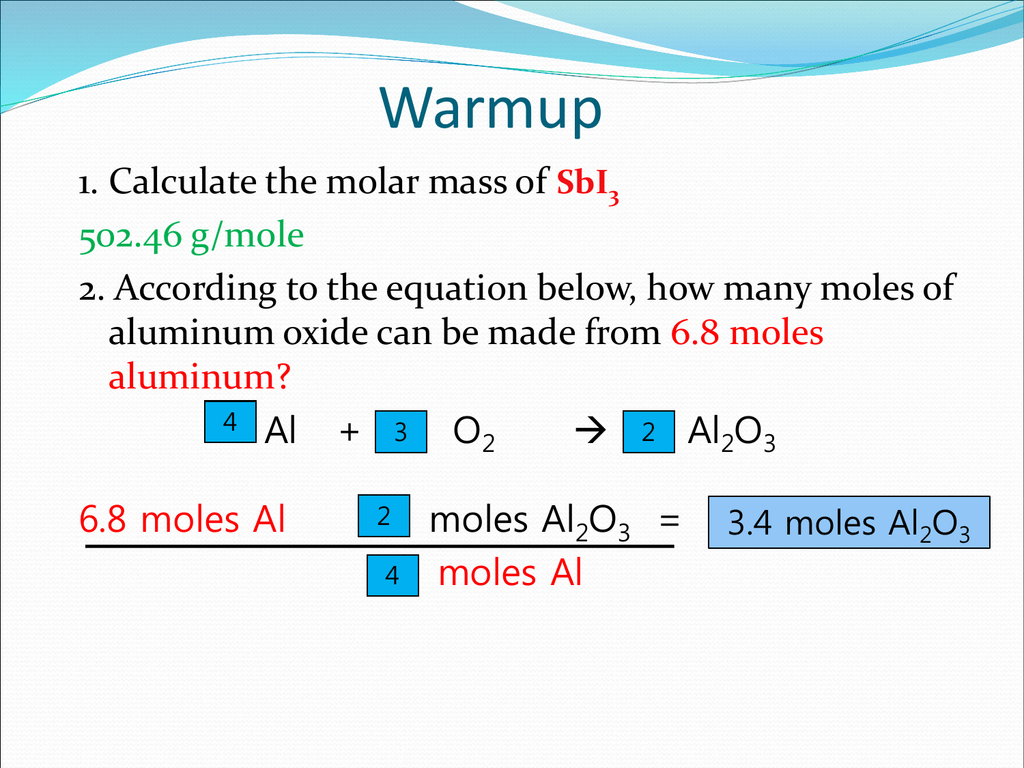
When some of the chemical formulas are not known, an experiment must be conducted to help determine the mole ratios. The compound with molecules containing the greatest number of atoms of N and O has the greatest molar mass. Solution This chemical equation is balanced, so the molar ratio of reactants and products may be used. The mass of a sample of any compound can be calculated by n×Mn×M, where n is the number of moles of molecules in the sample and MM is the molar mass (mass per mole) of the compound. A balanced chemical reaction equation gives the mole ratios of the reactants and the products as coefficients. Answer 1.81 mol N 2 O 4 Mole Relations Problem 2 Determine the number of moles of N 2 produced for the reaction 2 N 2 H 4 (l) + N 2 O 4 (l) 3 N 2 (g) + 4 H 2 O (l) when the reaction begins with 1.24 moles of N 2 H 4.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
